Business
Pune Metro Phase-2 Extension: Cabinet Approves Kharadi–Khadakwasla Line 4, Nal Stop–Warje–Manik Baug Line 4A – Stations, Length, Other Details
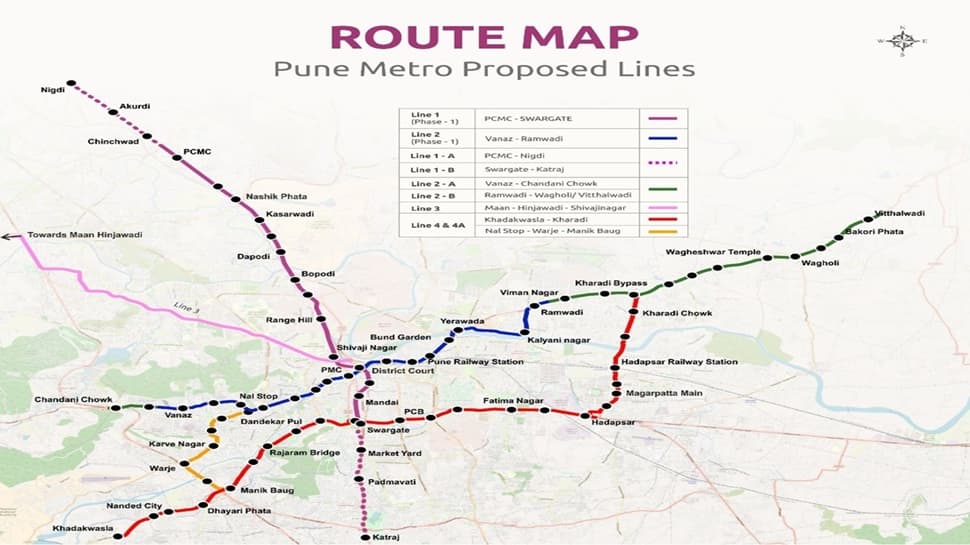
In a big boost for Pune’s local infra, the Union Cabinet has approved extension of Pune Metro’s Line 4 and Line 4A. Pune is set for another major boost in its public transport network as the Union Cabinet chaired by Prime Minister Narendra Modi, has approved Line 4(Kharadi–Hadapsar–Swargate–Khadakwasla) and Line 4A (Nal Stop–Warje–Manik Baug) under Phase-2. This is the second major project approved under Phase-2, following the sanction of Line 2A (Vanaz–Chandani Chowk) and Line 2B (Ramwadi–Wagholi/Vitthalwadi).
“Together spanning 31.636 km with 28 elevated stations, Line 4 and 4A will connect IT hubs, commercial zones, educational institutions, and residential clusters across East, South, and West Pune. The project will be completed within five years at an estimated cost of Rs 9,857.85 crore, to be jointly funded by the Government of India, Government of Maharashtra, and external bilateral/multilateral funding agencies,” said the Union Cabinet in a statement.
These lines are a vital part of Pune’s Comprehensive Mobility Plan (CMP) and will seamlessly integrate with operational and sanctioned corridors at Kharadi Bypass & Nal Stop (Line 2), and Swargate(Line 1). They will also provide an interchange at Hadapsar Railway Station and connect with future corridors towards Loni Kalbhor and Saswad Road, ensuring smooth multimodal connectivity across metro, rail, and bus networks.
From Kharadi IT Park to Khadakwasla’s scenic tourist belt, and from Hadapsar’s industrial hub to Warje’s residential clusters, Line 4 and 4A will knit together diverse neighbourhoods. Traversing Solapur Road, Magarpatta Road, Sinhagad Road, Karve Road, and the Mumbai–Bengaluru Highway, the project will ease congestion on Pune’s busiest routes while improving safety and promoting green, sustainable mobility.
According to projections, the daily ridership on Line 4 and 4A combined is expected to be 4.09 lakh in 2028, rising to nearly 7 lakh in 2038, 9.63 lakh in 2048, and over 11.7 lakh in 2058. Of this, the Kharadi–Khadakwasla corridor will account for 3.23 lakh passengers in 2028, growing to 9.33 lakh by 2058, while the Nal Stop–Warje–Manik Baug spur line will rise from 85,555 to 2.41 lakh passengers over the same period. These projections highlight the significant growth in ridership expected on Line 4 and 4A over the coming decades.
Cabinet approved expansion of Pune Metro Rail Project (₹9,858 Cr | 32 km)
Line 4: Kharadi – Khadakwasla (25.5 km, 22 stations)
Line 4A: Nal Stop – Manik Baug (6.1 km, 6 stations)Will ease traffic, reduce pollution, and improve commute within the city
Pune Metro… pic.twitter.com/ng6w75cmIJ
— Ashwini Vaishnaw (@AshwiniVaishnaw) November 26, 2025
The project will be implemented by Maharashtra Metro Rail Corporation Limited (Maha-Metro), which will carry out all civil, electrical, mechanical, and systems works. Pre-construction activities such as topographical surveys and detailed design consultancy are already underway.
With this latest approval, Pune Metro’s network will expand beyond the 100 km milestone, a significant step in the city’s journey towards a modern, integrated, and sustainable urban transit system.
With Line 4 and 4A, Pune will not just get more metro tracks, it will gain a faster, greener, and more connected future. These corridors are designed to give back hours of commuting time, reduce traffic chaos, and provide citizens with a safe, reliable, and affordable alternative. In the years ahead, they will emerge as the true lifelines of Pune, reshaping urban mobility and redefining the city’s growth story.
Business
Intellia Therapeutics says its Crispr-based treatment succeeds in pivotal trial

Intellia Therapeutics, building exterior and company sign, Cambridge, Massachusetts, USA.
Spencer Grant | Universal Images Group | Getty Images
Intellia Therapeutics said its Crispr-based treatment for a rare swelling condition met its goals in a late-stage trial, marking a milestone for the field of gene editing and putting the company on track to seek approval from the U.S. Food and Drug Administration.
The company’s treatment uses Nobel Prize-winning technology Crispr to edit DNA and turn off the gene that controls production of a peptide that’s overactive in people with hereditary angioedema, causing them to experience potentially life-threatening swelling attacks. Intellia’s treatment is administered once through an hourslong infusion, making the edits directly in the liver.
Intellia said the one-time treatment reduced attacks by 87% compared with a placebo, meeting the study’s main goal. Six months after treatment, 62% of patients were free from attacks and weren’t using other therapies, Intellia said.
The company described the safety and tolerability of the treatment as “favorable,” reporting the most common side effects were infusion-related reactions, headaches and fatigue. Analysts were closely watching safety in the trial since a patient in a separate trial of a different treatment from Intellia died. That patient developed a liver injury and ultimately died from septic shock following an ulcer, according to the company.
“When you think about where we started with Crispr, just 12 years ago with some of the fundamental insights, I think there was a lot of talk about what might be possible, and we’ve had reports along the way in terms of milestones, but this is the first Phase 3 data in any indication with in vivo Crispr where you’re actually changing a gene that causes disease,” said Intellia CEO John Leonard.
The only FDA-approved Crispr-based medicine comes from Vertex Pharmaceuticals. Called Casgevy, the gene editing is done outside the body, or ex vivo. The process requires collecting a person’s blood cells, making the edits outside the body, then reinfusing them back into a patient. Intellia’s treatment, meanwhile, makes the edits inside the body, or in vivo.
Intellia said it has started a rolling application with the FDA and plans to complete the filing in the second half of this year. The company expects to launch the treatment in the U.S. in the first half of next year, if it’s approved.
If approved, Intellia’s treatment, lonvoguran ziclumeran, will compete with about a dozen other chronic drugs for HAE. Despite the allure of a one-time treatment, genetic medicines haven’t always been a commercial successes. BioMarin withdrew its gene therapy for Hemophilia A because of weak sales, for example.
Leonard said there are important differences between the two, like the fact that BioMarin’s therapy faced questions about how long the effects would last. In contrast, he said Intellia hasn’t seen a single case in almost six years where the effects diminished over time.
Despite the results, he’s reluctant to call Intellia’s treatment a functional cure.
“I think this is a tipping point for the disease and tipping point for Crispr-based in vivo therapy where you can make a change [and] it’s permanent,” Leonard said. “And, as far as we can tell, we don’t have a single patient in this program or other program where there’s been any waning of the effect of what we did to the gene or the effect of what we’ve seen with the clinical aspects of the disease itself. So it’s pretty exciting.”
Clarification: This story has been updated to clarify that a patient in a separate trial of a different treatment from Intellia developed acute liver injury and ultimately died from septic shock following an ulcer.
Business
European flight prices are falling in short-term, Wizz Air boss says

While many airlines say they are raising prices due to high fuel costs, József Váradi says European airlines are trying to boost demand
Source link
Business
Claire’s closes all 154 stores in UK and Ireland with loss of 1,300 jobs

All of the chain’s standalone stores have stopped trading in the UK and Ireland.
Source link
-

 Sports1 week ago
Sports1 week agoNCAA men’s gymnastics championship: All-time winners list
-

 Sports1 week ago
Sports1 week agoWWE WrestleMania 42 Night 2: Live match results and analysis
-

 Politics7 days ago
Politics7 days agoUK’s Starmer seeks to deflect blame over Mandelson appointment
-

 Fashion1 week ago
Fashion1 week agoUK’s Sosandar returns to profitability amid robust FY26 performance
-

 Entertainment1 week ago
Entertainment1 week agoLee Anderson, Zarah Sultana kicked out of UK Parliament for calling PM ‘liar’
-

 Business1 week ago
Business1 week agoNo fuel shortage: Govt assures 100% domestic LPG, PNG, CNG supply amid Hormuz energy crunch – The Times of India
-

 Business1 week ago
Business1 week agoHow Trump’s psychedelics executive order could unlock stalled cannabis reform
-

 Sports1 week ago
Sports1 week agoQuetta Gladiators opt to bowl after winning toss against Peshawar Zalmi in PSL 11 clash






