Business
Eli Lilly’s weight loss pill orforglipron clears its latest trial, paving way for approval

Eli Lilly Biotechnology Center is shown in San Diego, California, U.S. March 1, 2023.
Mike Blake | Reuters
Eli Lilly on Tuesday said its daily weight loss pill helped patients with obesity and Type 2 diabetes shed weight in a late-stage trial, meeting the study’s main goal and clearing the way for the company to file for approval of the drug globally.
The treatment is inching closer to becoming a new, needle-free alternative in the highly lucrative market for weight loss and diabetes drugs called GLP-1s. More convenient pills could boost supply for the treatments and make them easier to access than the pricey weekly injections currently dominating the space. The Eli Lilly pill also would not come with dietary restrictions, in contrast to a similar oral treatment from chief rival Novo Nordisk.
Eli Lilly shares climbed more than 4% Tuesday.
The highest dose of the pill, orforglipron, helped patients lose 10.5% of their weight, or 22.9 pounds, on average at 72 weeks, compared to 2.2% weight loss among those who took a placebo. The drug’s weight loss in the study was 9.6% when analyzing all patients regardless of discontinuations.
Eli Lilly’s pill met the trial’s other goals of helping patients lower their hemoglobin A1c, a measure of blood sugar levels. By the end of the study, most patients no longer met the criteria for Type 2 diabetes based on that metric. The rate of side effects and treatment discontinuations in the study, called ATTAIN-2, appeared to be generally consistent with two recent phase three trials on Eli Lilly’s drug.
Eli Lilly said it now has the full clinical trial data package required to file for approvals of the drug for chronic weight management with global regulators. The pharmaceutical giant expects to launch the pill around the world “this time next year,” CEO David Ricks told CNBC in early August.
In an interview, Eli Lilly’s Chief Scientific Officer Daniel Skovronsky, said the pill had “unprecedented efficacy” in patients with obesity and Type 2 diabetes, who typically have a more difficult time losing weight compared to those without diabetes. Skovronsky said he hopes that diabetes patients will be able to use the pill earlier in their disease to slow down its progression.
Existing GLP-1 injections have shown greater weight loss than Eli Lilly’s pill, but having an oral option that offers more than 10% for patients with both obesity and Type 2 diabetes is “actually very good, so that’s positive,” said Dr. Caroline Apovian, co-director of the Center for Weight Management and Wellness at Brigham and Women’s Hospital.
But she called it “concerning” that more patients – 10.6% of those on the highest dose – stopped taking the pill due to side effects compared with what has been seen in separate studies on the existing injections, Eli Lilly’s Zepbound and Novo Nordisk‘s Wegovy. Apovian said patients and their prescribers will have to weigh the risks and benefits of the pill and the injections when deciding which to take, including convenience, side effects and efficacy.
The side effects of Eli Lilly’s pill were mainly gastrointestinal, such as nausea and vomiting, and were mild to moderate in severity. An estimated 23.1% of those who took the highest dose experienced vomiting, while 36.4% and 27.4% had nausea and diarrhea, respectively.
Around 20% of patients stopped taking the pill for any reason, which is roughly the same rate as the placebo group. Skovronsky said that reflects a combination of reasons apart from side effects. Those can include patients who wanted to stop participating in a trial because they were able to access another obesity medication, or those who didn’t lose enough weight because they were taking a lower dose of the drug.
But he said most patients stay on the drug, and said “the most important thing here is the scale of the opportunity,” referring to the number of people around the world could benefit from orforglipron. More than 100 million adults in the U.S. alone have obesity, according to Centers for Disease Control and Prevention data.
Apovian said she’s most excited about an oral option entering the market not because of how much weight loss it can promote, but rather its potential to expand access to obesity treatments, especially since a pill is much easier to manufacture than an injection. Apovian hopes Eli Lilly’s pill will be priced lower than injections — which cost roughly $1,000 per month before insurance — and receive broader insurance coverage.
Tuesday’s results are the third set of late-stage data the company has released on orforglipron this year. In April, the pill succeeded in a shorter phase three trial on diabetes patients without obesity.
Earlier this month, the drug also met the goals of a separate study on patients with obesity and not diabetes, but fell short of Wall Street’s expectations. The pill’s weight loss was higher in that trial compared to Tuesday’s data, but “that’s expected,” given the differences in patient populations, said Dr. Jaime Almandoz, medical director of the Weight Wellness Program at UT Southwestern Medical Center.
Overall, some doctors have touted the pill’s weight loss in the trials, and some analysts say it will still be a viable competitor in the space due to factors such as its easier manufacturing and lack of dietary restrictions.
“The convenience and specifically the lack of fasting requirements and water restrictions with orforglipron is likely one of the main differentiators when people think about other oral agents with similar effectiveness for weight change and A1C levels,” said Almandoz.
He said a pill creates additional opportunities for individualized patient care “where there’s more autonomy.”
For example, some people may be hesitant to take injections for weight loss, said Dr. Andrew Krafston, clinical associate professor at the division of metabolism, endocrinology and diabetes at the University of Michigan
“While this is a surmountable barrier for many, oral therapy aligns with traditional medication treatment and may have higher acceptability,” he said, adding that pills are less likely to be in short supply.
Detailed trial data
The trial followed more than 1,600 people, who were randomized to receive three different target doses of Eli Lilly’s pill or a placebo. Patients started at a lower dose of the drug and gradually increased it at four-week intervals to reach their final target dose.
More than 50% of patients on the highest dose of Eli Lilly’s treatment lost at least 10% of their weight in the trial, while 28.4% of those participants lost at least 15%. Across the different groups, the company did not disclose how many patients lost at least 5% of their weight.
Orforglipron “may not be the answer” for patients who are morbidly overweight, said Howard Weintraub, clinical director of the Center for the Prevention of Cardiovascular Disease at NYU Langone Heart. But he said, “for a lot of people who need to lose a fair amount of weight, losing 10% can make a big diference.”
Orforglipron lowered A1C by an average of 1.3% to 1.8% across different doses at 72 weeks, from a starting level of 8.1%. Around 75% of participants taking the highest dose achieved an A1C of 6.5% or less, which is at or below what the American Diabetes Association defines as diabetes.
Eli Lilly’s pill also improved key cardiovascular risk factors.
“We now have study after study with GLP-1 agonists showing that they reduce very important outcomes, which is what I do every day as a preventive cardiologist,” Weintraub said. “I try my very best to prevent them from having heart attack, stroke and death.”
Eli Lilly’s pill works in a similar way to Wegovy, Ozempic and Novo Nordisk’s diabetes pill Rybelsus, targeting a gut hormone called GLP-1 to suppress a person’s appetite and regulate blood sugar. Novo Nordisk is also seeking approval of an oral version of Wegovy, which could come by year-end.
But unlike those three medications, Eli Lilly’s pill is not a peptide medication. That means it is absorbed more easily by the body and does not require dietary restrictions like Rybelsus or oral Wegovy does.
Business
US stocks today: Wall Street inches higher as markets eye ceasefire deadline; Dow jumps 300 points, S&P 500 remains flat – The Times of India

US stocks moved higher on Tuesday, as investors remained optimistic over a possible extension of the US-Iran ceasefire. Markets showed early strength, with the Dow Jones Industrial Average rising 0.56% or 279 points to 49,721.56 around 8 pm IST. The S&P 500 inched up 0.2% to 7,129, while the Nasdaq Composite gained 96 points or 0.4% to reach 24,500. As trading progressed, the upward momentum strengthened, with the Dow climbing 397 points, or 0.8%, and the S&P 500 adding 0.2%, putting it within reach of another record high. The Nasdaq remained modestly higher. Investor sentiment was shaped in part by developments in the Middle East. Oil prices, which had surged a day earlier amid renewed disruption to the Strait of Hormuz, eased on Tuesday. Brent crude slipped 0.7%% to $94.78 per barrel ahead of the expected expiry of a two-week ceasefire between the United States and Iran. The conflict has driven sharp swings in oil markets, with prices ranging from about $70 before the war to peaks of $119 as concerns over a prolonged closure of the key shipping route intensified. Economic data released during the session pointed to continued resilience in consumer activity. US retail sales rose 1.7% from the previous month to $752.1 billion, beating expectations, largely due to higher petrol prices. Spending remained relatively steady even when excluding gasoline sales, indicating broader stability in consumption during the first full month of the conflict. Global markets presented a mixed picture, with European indices trading unevenly after a stronger performance in Asia, where South Korea’s Kospi index jumped 2.7%. In the bond market, US Treasury yields edged higher, with the 10-year yield ticking up to 4.27% from 4.26% the previous day. Attention is also turning to Washington, where Kevin Warsh, nominated by US President Donald Trump to lead the Federal Reserve, is scheduled to testify before Congress later in the day. Investors are expected to closely watch his remarks for indications on interest rate policy and the central bank’s independence.(Disclaimer: Recommendations and views on the stock market, other asset classes or personal finance management tips given by experts are their own. These opinions do not represent the views of The Times of India.)
Business
Leave, holidays and encashment: What India’s changing labour laws mean for employees – The Times of India

Leave is often seen as a simple workplace benefit – an approved absence from work. In reality, it is one of the more structured and regulated aspects of employment in India. With the implementation of new labour codes, questions around leave entitlement, holidays and leave encashment have drawn renewed attention. This matters because these rules affect not just everyday working life, but also what happens when an employee leaves an organisation.For employers and employees, understanding how leave works today is not always straightforward. This is because two legal systems operate side by side: the new central labour codes and the older State-level Shops and Establishments (S&E) laws. While the intent is to move towards a simpler and more uniform system, the actual position still depends on job role, location and which law applies.Different types of statutory leaveIndian labour laws recognise several types of statutory leave. The most important is earned leave (also called privilege leave). This leave builds up over time based on how many days an employee works. In addition, there are provisions for sick leave, casual leave, and national and festival holidays.Earned leave is different from other types of leave because it has both time-off value and financial value. If it is not used, it can build up and may be paid out in cash – either during employment or when the employee leaves, subject to carry forward limits – depending on the applicable law and company policy.Sick leave and casual leave, on the other hand, are meant for short-term or urgent needs and are usually not designed to be encashed.National and festival holidays form a separate category. These ensure paid holidays on important national or regional days, based on State notifications and local rules.Labour codes vs Shops and Establishments lawsA frequent point of confusion is the interface between the labour codes and State Shops and Establishments Acts.The Occupational Safety, Health and Working Conditions Code introduces a common framework for leave, but for people classified as “workers” under that law. At the same time, State S&E laws continue to apply to many salaried employees working in offices, shops and service-sector businesses.Because of this, uniformity has not fully arrived yet. Different State laws and leave rules may still apply for employees depending on where they are employed and work. Those who fall under the labour code framework move towards a more standard national system. Where both laws could apply, guidance from authorities suggests that the more beneficial provision would generally continue to apply.

Employers are expected to apply these frameworks together and ensure consistency as the new system takes shape.How earned leave builds upEarned leave generally depends on how long an employee has worked.Under the labour codes, earned leave accrues at a standard rate of one day for every twenty days of work, subject to certain eligibility conditions. This is meant to create a common reference point across the country.State Shops and Establishments laws, however, follow different approaches. Some States grant a fixed number of leave days each year, while others link leave closely to days worked. States also differ on how much unused leave can be carried forward.Sick leave, casual leave and holidaysSick leave and casual leave are mainly meant for short-term protection rather than long-term accumulation. Sick leave helps employees during illness, while casual leave allows flexibility for sudden personal needs.These types of leave are mostly governed by State law and internal company policy, with limited direct impact from the labour codes. Usually, unused sick or casual leave does not carry forward.National and festival holidays are largely decided at the State level. Employers are expected to follow notified holiday lists or compensate employees who work on those days, as per State rules.Carrying forward unused earned leaveHow unused earned leave is treated is one area where the labour codes bring more structure.Earlier, State laws allowed different levels of leave accumulation. Under the labour code approach, carry-forward is subject to clear limits, after which settlement mechanisms may apply. This is intended to avoid unlimited build-up of leave while still protecting employee interests.If leave could not be taken because of work requirements, safeguards exist to ensure such leave is not lost automatically.Annual leave encashment under labour codesAnother change under the labour codes is clearer recognition of leave encashment during ongoing employment.Earlier, in many States, leave was typically encashed only when an employee resigned, retired or was terminated. Under the new labour codes framework, employees may be entitled to encash leave exceeding permissible carry forward limits even while they remain in service. As per provisions under labour codes, a worker shall be entitled on his / her demand for encashment of leave at the end of calendar year. Worker shall be entitled, where the total number of leave exceeds 30 days, to encash such exceeded leave.Leave encashment when employment endsAcross Indian labour laws, one position has remained largely consistent. Unused earned leave is expected to be settled when employment comes to an end, whether the employee resigns, retires, is retrenched or is terminated.How this amount is calculated depends on the applicable law. State S&E laws refer to specific wage definitions, while the labour codes require calculation using the definition of “wages” under the Code. This may differ from earlier practice.
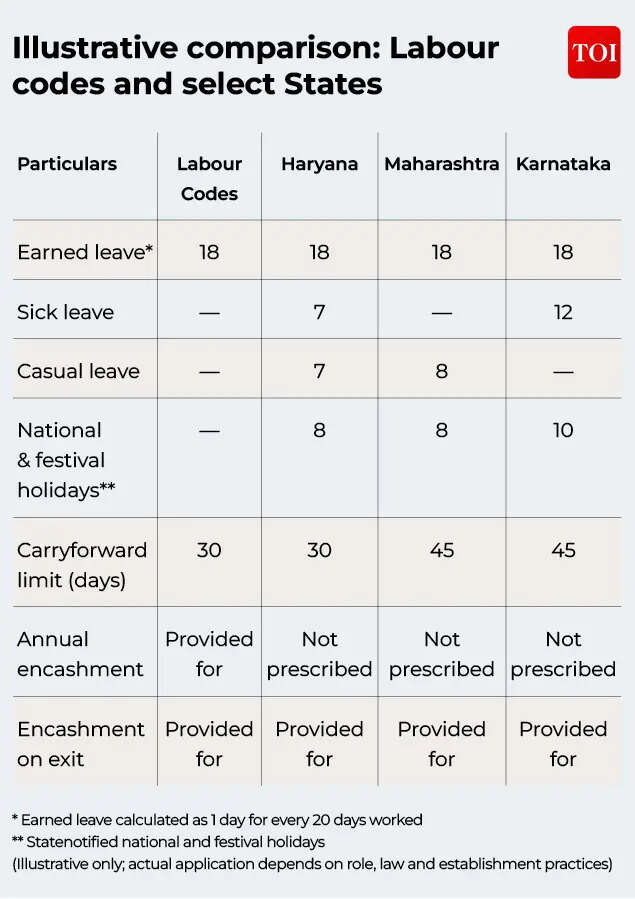
What employees and employers should keep in mindFor employees, the key point is that leave is not only a company benefit but part of a legal framework. How it applies depends on role, location and legal coverage.For employers, the focus remains on aligning internal policies with both Central and State laws, while ensuring smooth implementation. Clear communication and regular policy reviews will continue to be important during this transition.Leave rules may not attract the same attention as pay or job security, but they play a quiet role in work-life balance and financial certainty. As India’s labour framework evolves, earned leave is increasingly seen not just as time away from work, but as a regulated employment benefit with defined outcomes.(The author, Puneet Gupta is Partner, People Advisory Services Tax at EY India)
Business
Electricity bills targeted in planned shakeup to energy pricing

The war in the Middle East has brought renewed attention to Britain’s vulnerability to energy price shocks.
Source link
-

 Fashion5 days ago
Fashion5 days agoFrance’s LVMH Q1 revenue falls 6%, shows resilience amid Iran war
-

 Sports1 week ago
Sports1 week agoThe case for Man United’s Fernandes as Premier League’s best
-

 Entertainment1 week ago
Entertainment1 week agoPalace left in shock as Prince William cancels grand ceremony
-

 Business1 week ago
Business1 week agoUK could adopt EU single market rules under new legislation
-
 Entertainment6 days ago
Entertainment6 days agoIs Claude down? Here’s why users are seeing errors
-

 Fashion1 week ago
Fashion1 week agoEnergy emerges as biggest cost driver in textile margins
-

 Business1 week ago
Business1 week agoDelta Air Lines unveils first new Delta One suite in premium cabin arms race
-

 Fashion1 week ago
Fashion1 week agoAsia claims largest share of markets on Kearney FDI Confidence Index











